The Resources page offers some multimedia content such as our brochure, Q&A and video gallery, and some technical resources related to the mRNA Programme.









Q&A
Technology transfer is a logical procedure that controls the transfer of any process together with its documentation and professional expertise between development and manufacture or between manufacture sites. It is a systematic procedure that is followed in order to pass the documented knowledge and experience gained during development and/or commercialisation to an appropriate, responsible and authorised party.
Technology transfer embodies both the transfer of documentation and the demonstrated ability of the receiving unit (RU) to effectively perform the critical elements of the transferred technology, to the satisfaction of all parties and any applicable regulatory bodies.
Typically, technology transfer is a bilateral process – from the originator of the technology to a single recipient. With regards to the mRNA technology transfer programme a unique model using a multilateral process will be implemented – from the originator of the technology to multiple recipients.
A technology transfer hub refers to a technology donor that develops a technology and then transfers it multilaterally (single donor, multiple recipients). The Hub brings together in one single place all the elements needed to develop the technology (know-how, data, intellectual property) and then transfers the manufacturing process to multiple technology recipients.
Multilateral technology transfer is needed when there is a request from multiple recipients to receive the technology and when there is limited interest in bilateral agreements.
A technology donor is an institution who owns know-how, data and/or intellectual property rights related to a technology, for example a manufacturing process or a product composition or design and that agrees to transfer it.
A technology recipient is an institution who receives the documented experience, knowledge and, in some instances, intellectual property rights related to a technology from a donor. The technology recipient may or may not have prior experience and expertise related to the technology being transferred, for example, a manufacturing process.
As with other forms of outbreaks, the COVID-19 pandemic, has underscored that gross inequity exists in access to health products, especially vaccines.
During the pandemic, low- and middle-income countries (LMICs) have been left highly vulnerable as most advanced technologies and vaccine manufacturing capacity remained concentrated in a small number of highincome countries. Access was hindered further as vaccine rollout is often subjected to strong nationalism or regionalism during health emergencies.
A more widely distributed access to technologies and vaccine manufacturing network would reduce the gap in making vaccines available promptly for everyone, everywhere and accelerates the impact of the necessary collective effort to control outbreaks.
WHO has a critical role in coordinating such efforts, ultimately empowering countries to develop coherent health and industrial policies for sustainable investments towards pandemic preparedness.
By early 2021, it was clear that low- and middle-income countries would be pushed back to the end of the queue for receiving COVID-19 vaccines. This was due to production and supply constraints, to vaccine hoarding by wealthy countries and to companies prioritising sales to governments that could pay the highest price. While WHO continuously advocated for equitable sharing of vaccines through the COVAX Facility, and for sharing of
technology through bilateral and multilateral agreements (e.g. through the COVID-19 Technology Access Pool), the initiative of establishing an mRNA technology transfer programme based on a technology donor responsible to develop and transfer the technology to multiple recipients surfaced as a valid strategy to increase mRNA vaccine production capacity in under-served regions, and thus promote regional health security.
The mRNA Technology Transfer Programme aims to contribute to equitable access to mRNA vaccines by increasing the distribution of sustainable manufacturing capacity across LMICs, enhancing regional and inter-regional collaborations, and developing and empowering a local workforce through tailored and inclusive training and expert support.
The “mRNA Technology Transfer Programme” is convened by WHO and its partner Medicines Patent Pool (MPP).
MPP has extensive experience and knowledge on licensing, intellectual property, fund-raising and funds management, which are key areas in which MPP supports the Programme management.
In addition, MPP has recently created a Technology Transfer Team with development, biomanufacturing and Chemistry, Manufacturing, and Controls (CMC) experience. This team is providing support to both the technology donor and technology recipients in the process of establishing, transferring out and onboarding the technology. This ensures a continued support from technology development to a successful transfer of the technology platform to the Programme Partners.
The Programme is co-convened by WHO and the Medicines Patent Pool under the Act-Accelerator/COVAX umbrella. The Programme is currently funded by European governments/institutions, Canada and African institutions/organisations and comprises the organisations mentioned below (February 2023).
South African consortium:
• Afrigen (technology development and transfer centre): technology platform development (using COVID-19 as proof of concept) up to Clinical trial Phase I/II scale and technology transfer to the technology recipients.
• Biovac: 1st technology recipient responsible for technology platform scale-up and validation (up to Clinical trial Phase III scale).
• South Africa Medical Research Council (SAMRC): research on next generation improved technologies (manufacturing process and vaccine composition) together with vaccine candidates identification and early development of target diseases relevant for LMICs. SAMRC coordinates research activities of different organisations in South Africa (South African mRNA Vaccine Consortium – SAMVAC):
• Antiviral Gene Therapy Research Unit (AGTRU-University of Witwatersrand (WITS))
• The Genomic Surveillance Network of South Africa (NGS-SA)
• Kwazulu-Natal Research Innovation and Sequencing Platform (KRISP)
• Centre for Epidemic Response and Innovation (CERI-Stellenbosch University)
• African Centre of Excellence for Genomics of Infectious Diseases (ACEGID)
• Centre for Bioprocess Engineering Research (CeBER, University of Cape Town (UCT)), and the DSI Preclinical Drug Development Platform (PCDDP, North-West University).
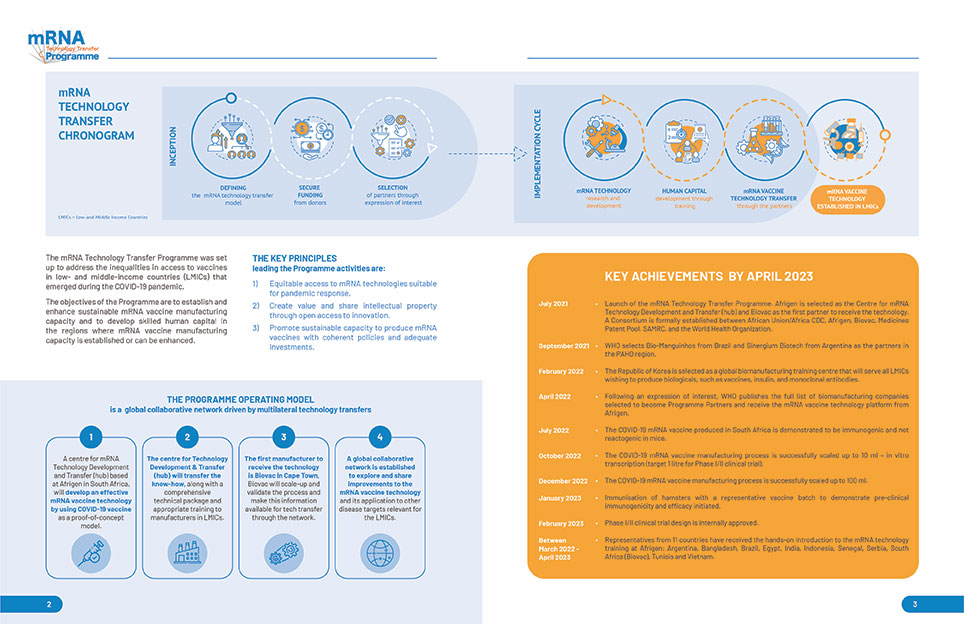
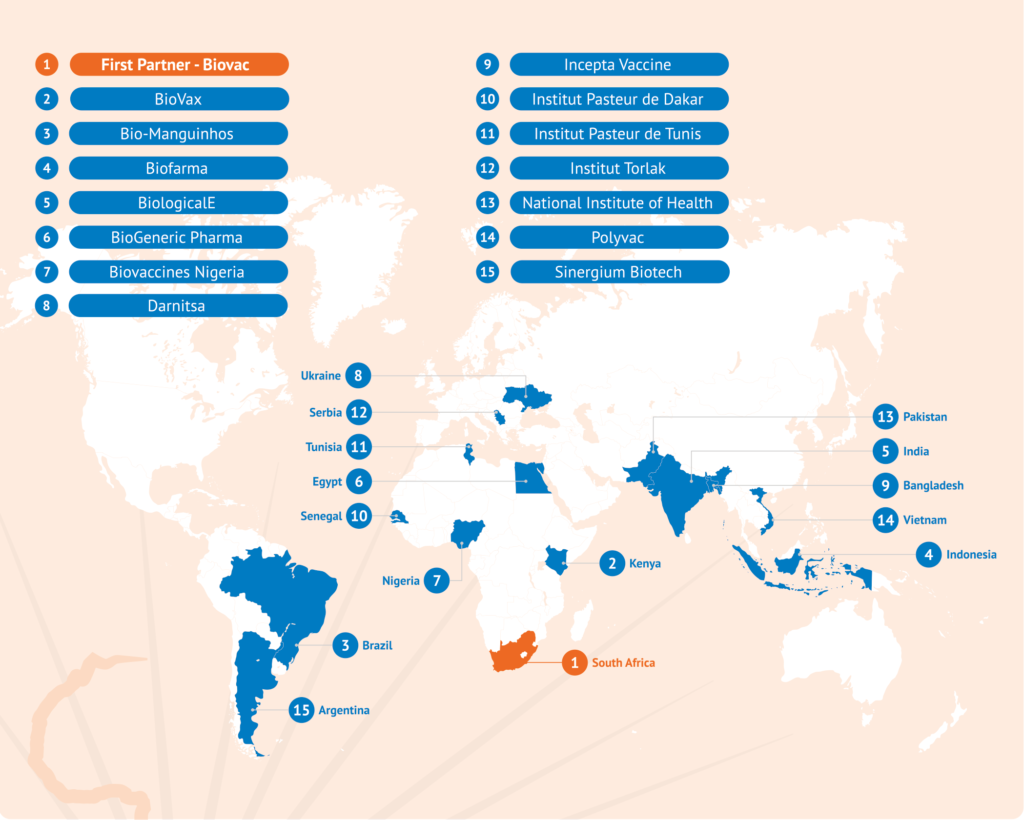
Network Partners: 15 technology receiving units from different regions in the world have been nominated by WHO (see picture below).
In April 2021, WHO issued a call for expressions of interest to companies wishing to host an mRNA technology transfer hub. In June 2021, WHO announced the selection of the South African consortium to establish the mRNA hub, which will serve as technology donor for all low- and middle-income countries.
Afrigen has established a non-GMP COVID-19 vaccine manufacturing process at laboratory scale and is currently scaling up the process to a scale suitable for manufacturing a vaccine batch to be used in a Phase I/II clinical trial in a GMP environment. Technology transfer to the network partners will happen in parallel.
As of February 2023, 15 partners in 15 different countries have been selected and announced by WHO.
Biovac, in South Africa, has been selected by WHO as the first recipient. They are part of the South African consortium also including Afrigen (the technology development and transfer centre) and SAMRC (research organisation) and will also scale up and validate the platform received.
Sinergium in Argentina and Bio-Manguinhos in Brazil were nominated by PAHO in September 2021. The other 12 partners were nominated by WHO in February-April 2022 as a result of an EOI opened in November 2021 (see also Q8 above).
Afrigen Biologics, the technology development and transfer centre will develop the technology platform to produce a COVID-19 vaccine to a scale suitable for a Phase I/II clinical trial. The technology platform developed by Afrigen will be transferred to Biovac that will also scale-up and validate it (using COVID-19 as a proof of concept disease target) and manufacture vaccine batches suitable for Phase IIb/III clinical trials.
All selected recipients will have access to technology transfer documentation packages at different stages of the process development performed by Afrigen and Biovac.
Following technology transfer, the recipients will decide whether they have interest to further develop/commercialise a COVID-19 vaccine or apply the technology they received to other pathogens of interest. Once a vaccine has been successfully developed, the recipients will be able to increase the process scale to enable supply of mRNA vaccines to support local and regional requirements. Each recipient will need to seek regulatory approval for the vaccine in their jurisdiction; for COVID-19 this will be facilitated by the availability of the full pre-clinical and clinical data generated by the technology donor and Biovac.
Following the signature of the Technology Transfer Agreement, MPP, together with WHO, will engage individually with each recipient to define the Technology Transfer Strategy. The recipients’ strategic intent and business plans will guide their technology transfer strategy considering both future pandemic readiness and inter-pandemic sustainability.
Technology transfer Package 1: Access to GMP mRNA technology. Enables the recipient to develop mRNA expertise at a scale commensurate with early-stage clinical trial material supply. This is suitable for partners intending to implement an internal platform for new vaccines development (up to Phase I/II).
Technology transfer Package 2a: Access to a scaled-up technology platform. Enables partners to implement an mRNA technology platform at a commercial scale applicable for COVID-19 and/or other disease targets.
Technology transfer Package 2: Access to scaled-up validated technology platform for a COVID-19 mRNA vaccine (including demonstration of process robustness and product quality consistency). Enables the recipient to leverage the validation of the technology platform at commercial scale to support validation in their own facility. This is applicable to the production of COVID-19.
Technology transfer 3: Access to mRNA COVID-19 Vaccine Marketing Authorisation Application documents. This enables the recipient to have access to documentation packages submitted for authorisation for commercialisation of the mRNA COVID-19 Vaccine in support of the recipients’ own national regulatory requirements and clinical pathway.
To develop the Afrivac2121 COVID-19 vaccine, the South African Consortium is using publicly available information regarding the composition of the Moderna Spikevax COVID-19 vaccine (mRNA sequence and lipids composition) and is independently developing the manufacturing process.
Moderna has publicly stated that they will not enforce patent rights for COVID-19 in the 92 LMICs belonging to the list of countries eligible to get access to COVID-19 vaccines through Gavi COVAX Advance Market
Commitment.
The Consortium, in collaboration with South African academic groups and biotech companies outside of South Africa are developing next generation mRNA vaccine technologies. Intellectual property resulting from this activity will be held by the inventors but will be made freely available to recipients to the mRNA Technology Transfer Programme.
The Medicines Patent Pool (MPP) – which co-leads the Programme initiatives with WHO – will assist with its expertise in intellectual property management by providing intellectual property analysis and defining and
negotiating terms and conditions of agreements that might have to be signed.
Freedom to Operate (FTO): MPP and WHO will not guarantee FTO at country level but will provide an Intellectual Property landscape analysis detailed at country level. The check of actual status of patents/claims filed and/or granted in the country is the recipient’s responsibility.
Intellectual Property sharing: Novel and inventive data generated by the recipient in the framework of the Programme and/or through data received from the Programme will be shared with other recipients in the Programme.
Third party Intellectual property: If recipients are granted access to third party IP, they have the obligation to undertake reasonable efforts to license that IP to MPP (and the Programme) only if the IP is linked to the research, development or commercialisation of a product partially or entirely based on the technology transferred.
There are several factors that make mRNA technology attractive to the Programme:
• mRNA vaccines have proved efficacious against COVID-19 caused by the ancestral SARS-CoV-2 Wuhan strain and against cross-reactive new variants.
• Though relatively new, the mRNA vaccine manufacturing technology platform is easy to share, develop and adapt to new variants.
• The mRNA technology platform developed using COVID-19 as proof of concept can potentially be adapted to vaccines for other diseases, with modest process adaptations. Investing in training for use of this technology will have long-term benefits for the regions where production will be based, most of which are currently under-served by the global pharmaceutical market.
• Due to the low mRNA dose required to elicit a human response, this technology allows for producing high numbers of doses at a relatively small scale, compared to other vaccine technologies, making it suitable for LMICs.
Numerous biotech companies have announced plans to establish mRNA vaccine production in LMICs. WHO welcomes all initiatives aiming to expand quality biomedical manufacturing in Africa and other under-served regions. These bilateral partnerships are good contributions to the overall effort and WHO stands ready to support all countries involved at their request.
Within the broader context of promoting local production, technology transfer alone is insufficient. Two major barriers need to be overcome – the low availability of a trained workforce and weak regulatory capacity.
WHO is addressing these gaps by establishing a biomanufacturing workforce training hub and working with the WHO Academy to ensure that regulatory and biomanufacturing training meet countries’ needs and objectives.
Regulatory systems play a key role in assuring the quality, safety, and efficacy of medical products and in licensing them so that they may be made available to populations. WHO is working with multiple regulatory authorities in countries that are participating in the mRNA technology transfer Programme by evaluating them through its global benchmarking tool (GBT).
The tool and benchmarking methodology enable WHO and regulatory authorities to identify strengths and gaps and build improvement plans. Once improvements have been made, the regulatory authority is assessed according to its maturity level on a scale that ranges from 1 (existence of some elements of a regulatory system) to 4 (operating at advanced level of performance and continuous improvement).
WHO intends to use the GBT to evaluate and publicly designate WHO-listed authorities (WLAs) that have been objectively documented to perform at maturity levels 3 and 4. Several countries involved in the Programme are at an advanced stage of being assessed for their maturity level.
While the initiative will contribute to regional capacity to produce vaccines, we have seen during the COVID-19 pandemic that supply chains are a critical barrier to national and regional responses. During the early phase of the pandemic, supplies of key materials for vaccine production, such as glass vials or rubber stoppers were not reaching regions lacking their own capacities to produce them. Acknowledging the challenge and the possibility this might become a barrier to local production, WHO is exploring opportunities to facilitate regional production of some of these materials.
No, Afrigen is developing its own COVID-19 mRNA vaccine (Afrivac2121). The composition of Afrivac2121 mimics the Moderna COVID-19 vaccine, according to publicly available information. The manufacturing process is being entirely designed at Afrigen.
Afrigen is conducting product and process development which will then be shared with 15+ recipients. If each recipient was to undertake the development independently, this would be a suboptimal use of precious resources, without considering that some would not have the capacity. By having the development done at one central site, all the recipients will have access to free know-how and established procedures without the need for investment in development.
The Programme aims at accelerating access to know-how and capacity building for the mRNA technology platform. Starting from R&D small scale, technology recipients can familiarise themselves with the technology. Additionally, the recipients can start building the competences needed to implement mRNA-based products and gain confidence at small scale on the procedures and essential parameters to prepare the next steps of technology transfer.
COVID-19 is the only disease for which mRNA vaccines are approved. As the Hub did not receive a technology transfer but is developing its own vaccine/manufacturing process, a proof of concept must be established by manufacturing a vaccine for which a reference product is available to demonstrate safety and efficacy.
While the Programme is developing its own mRNA technology using COVID-19 as a proof of concept disease target, the technology and respective manufacturing process and analytics can be applied to other disease targets. The composition of the mRNA-based vaccines will require careful assessment considering countryspecific Intellectual Property and Freedom To Operate. The R&D network will facilitate the establishment of next generation technologies focusing on lowering Cost of Goods (CoGs) and improving stability. In addition, the sharing of know-how within the Programme Network will support the development of other mRNA vaccines relevant to LMICs.
WHO does not procure vaccines but recipients are encouraged to seek WHO pre-qualification (PQ) of the vaccines they produce which is a requirement for the vaccines to be procured by international agencies (GAVI, UNICEF, Revolving Fund, etc.).
Latest newsAll News
The database of investments to support local COVID-19 vaccine production compiles detailed information about each COVID-19 vaccine candidate in development in countries where the mRNA technology transfer hub Programme has selected technology recipients.
In April 2021 the WHO announced the mRNA technology transfer Hub Programme to support the local production of vaccine doses. This announcement is part of a global effort to support local manufacturers. To help Programme our funders having to have a broader understanding of the current investment in COVID-19 vaccine local production, MPP has been asked to map out parallel initiatives(definition below). The database aims at improving equitable access and prepare countries for the next pandemic by providing an overview of the pandemic readiness plan of manufacturers in LMICs. Doing so will facilitate global coherence to prevent duplication of work and identify possible gaps. Moreover, the WHO encouraged collaboration with parallel initiatives to avoid duplications but also to provide support to some of them with access to training and support with regulatory aspects.
PARALLEL INITIATIVE (as used in this context): any project developed independently from the mRNA technology transfer Hub Programme and led by private or public partners that support pandemic preparedness with a specific focus on the local production of COVID-19 vaccines. MPP’s database lists exclusively investments made on continents where recipients of the technology (spokes) are located.
All data information is gathered via public sources including press releases, CHAI and UNICEF, data are cross-checked with multiple sources. The source of information is listed on the database, however, MPP does not take any responsibility for the accuracy of public information shared by vaccine providers or found online. MPP disclaim any liability or responsibility regarding data toward any vaccines’ quality, safety, efficacy, or merchantability.
The database is updated monthly if new information is available. We welcome your feedback to help us update or make necessary changes. For questions relating to the database please contact us.


















